
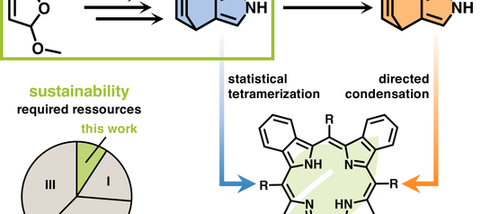
Sustainable Synthesis of 4,7-Dihydro-4,7-ethano-2H-isoindoles—Key Building Blocks to Benzoporphyrins
Chem. Eur. J. 2026 , e70979. Benzoporphyrins, π extended porphyrin analogues, exhibit remarkable potential for optoelectronic and theragnostic applications, yet have remained largely confined to fundamental studies owing to the challenging synthesis of masked isoindole precursors. Here, we report a concise, scalable three-step synthesis of 4,7-dihydro-4,7-ethano-2 H -isoindole, a key building block of the benzoporphyrin framework. Subsequent functionalization affords the co


From Yellow to Red: Emission Tuning of Benzothioxanthene Imides Through Selective Multi-Arylamine Functionalization
ChemPlusChem 2026 , 91 , e70135 . The selective bis- and tris-functionalization of benzothioxanthene imides (BTI) with extended triphenylamine (eTPA) units, enabling precise tuning of optical and electronic properties, is introduced herein. Controlled bromination and subsequent Suzuki–Miyaura cross-coupling allow modular introduction of donor groups, resulting in redshifted emission into the red–near-infrared (NIR) region with large Stokes shifts and fluorescence quantum yi


A Modified Pyrrole Biscarbinol Approach to Opp-A2B2-Symmetric Benzoporphyrins
ChemistryEurope 2026 , 4 , e202500297. In the pursuit of advanced functional materials, π-extended porphyrin derivatives, particularly β , β -benzannulated tetrapyrroles, have garnered significant attention due to their unique structural and electronic properties. This study refines existing methodologies for the selective synthesis of opp -A 2 B 2 -symmetric benzoporphyrins based on 4,7-dihydro-4,7-ethano-2 H -isoindole or [3,4]epipyrroloanthracene building blocks. By cond


























